Which of the Following Elements Has the Largest Ionization Energy
Thus helium has the largest first ionization energy while francium has one of the lowest. Element Q is a third period element and has the following successive ionization energies.
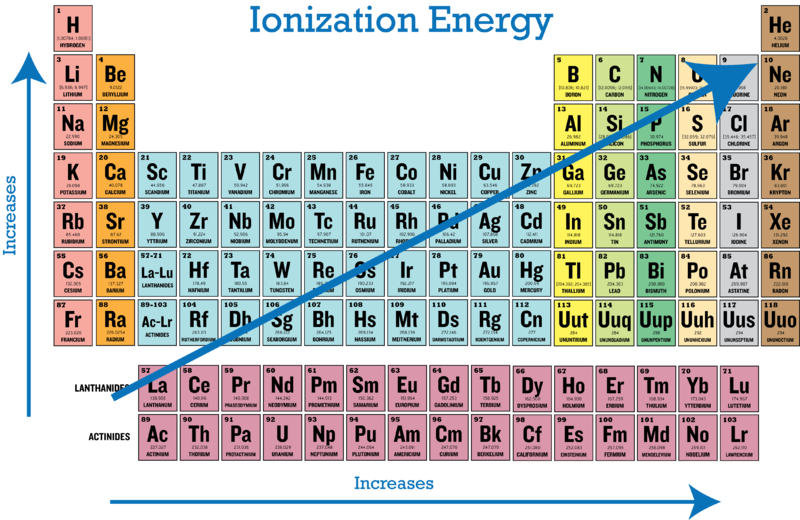
Ionization Energy Periodic Table Trends Chemtalk
Tramwayniceix and 57 more users found this answer helpful.
. Which of the following elements has the largest second ionization energy ie2. Element Q is a third period element and has the following successive ionization energies. 2nd IE 11454 kJ mol-1 For Sr.
Thus helium has the largest first ionization energy while francium has one of the lowestThus helium has the largest first ionization energy while franciumfranciumFrancium is a chemical element with the symbol Fr and atomic number 87. Which element has the largest ionization energy in its period. Ionization energy of Beryllium Be 932 eV.
As a chemist as a physical scientist you should however always examine the data. Which of the following elements has the largest third ionization energy IE3. IE1 IE2 IE3 IE4 IE5 IE6 577 kJmol 1816 kJmol 2744 kJ 11576 kJmol 14829 kJmol 18375 kJmol.
119 rows - Elements in human body. Its most stable isotope francium-223 originally. 60 - Covalenz radius.
That ionization energy should. Ionization energy decreases DOWN a Group and increases from left to right across a Period as we face the Periodic Table. 5441 eV 3.
The first ionization energy varies in a predictable way across the periodic table. Silicon A mixture of sawdust gravel sand and salt is placed in a beaker of water. Therefore potassium will have the highest ionization energy from the four elements given to you.
2nd IE 2633 kJ mol-1. Li has the largest second ionization energy IE2. Thus helium has the largest first ionization energy while francium has one of the lowest.
This problem has been solved. Ionization energy of Helium He 2458 eV. It is the energy required to ionize remove one electron at a time from the atoms gaseous state.
Which of the following element has highest third ionization enthalpy IE3. The ionization energy decreases from top to bottom in groups and increases from left to right across a period. Elements at the lowest points are Li Na K Rb and Cs.
It is extremely radioactive. The ionization energy decreases from top to bottom in groups and increases from left to right across a period. A Ca B Sr C Na D CIE C 11.
Which of the following elements would have the greatest difference between the first and the second ionization energies. When N a ions loses an electron this stable configuration of N e is broken. Out of the answer choices rubidium has the highest energy valence shell.
Yes The difference between 2 successive points are 8 and 18. Which of the following elements has the largest first ionization energy. Which element has the highest second ionization energy.
Which of the following elements has the largest second ionization energy IE2. When sodium loses one electron it forms N a ion which has stable electronic configuration of the noble gas N e. Elements at the highest points are He Ne Ar Kr and Xe.
4 rows The ionization energy decreases from top to bottom in groups and increases from left to right. With a single electron in the fifth energy level krypton will have the highest number of energy levels of the group I elements listed. Colorwhite2nd IE 10642 kJ mol-1 For K.
This requires high amount of energy. 11 rows If we were to take a single element then Helium is said to have the highest first ionization. Prior to its discovery it was referred to as eka-caesium.
The ionization energy increases from left to right of the periodic table. What is element Q. Which element listed below would most likely have the.
Colorwhite2nd IE 3052 kJ mol-1 For Rb. Of the following elements which one has the smallest first ionization energy. Ionization energy of Lithium Li 539 eV.
Sodium atoms have the largest second ionization energy. When moving across a period atomic radius will decrease as the number of protons increases. Which of the following elements has the largest first ionization energy.
The actual values are For Ca. Which of the following has the highest second ionization energy sodium or magnesium. B chlorine has a greater ionization energy than sodium does C chlorine has a greater electronegativity than sodium does D chlorine is a gas and sodium is a solid.
First Second and Third Ionization Energy eV First Ionization Energy. What is ionization energy. IE1 IE2 IE3 IE4 IE5 IE6 577 kJmol 1816 kJmol 2744 kJ 11576 kJmol 14829 kJmol 18375 kJmol.
Experts are tested by Chegg as specialists in their subject area. When Na ions loses an electron this stable configuration of Ne is broken. Who are the experts.
The first ionization energy varies in a predictable way across the periodic table. 1 Li 2 Ne 3 F 4 Be. A Be B Mg C Ca D Sr E Ba 10.
What is element Q. Hence second ionization energy of sodium is largest. The ionization energy decreases from top to bottom of the periodic table.
Which element listed below would most likely have the. Ionization energy of Hydrogen H 1359 eV 2. The ionization energy of atoms in the top right corner of the periodic table is higher because the electrons are in orbitals closer to the nucleus causing them to be harder to remove.
We review their content and use your feedback to keep the quality high. The order of ionization energies is thus smallest to largest. Which of the following elements has the largest second ionization energy IE2.
Here is a start. This requires high amount of energy. Hence second ionization energy of.

The Parts Of The Periodic Table

Comments
Post a Comment